Even when they aren't blowing up the countryside, explosives and rocket propellants can be harmful to the environment.
Ecological damage is expected in war, but as a side effect. Minimizing ecological damage is important for many reasons. First, after hostilities cease, people need to live, work, and farm in the areas previously engaged in conflict. There is also the effect that harmful or toxic compounds have on the troops using the munitions. Winds, water currents, and groundwater supply contamination can affect non-combatant nations, including allies and the nations using the munitions.
But even if there were no hostilities, the mere production of the munitions can have ecological consequences, as can the testing of them, and their use in training and target practice. Moreover, when bombs and shells age, or become obsolete, they must be disposed of in a way that does not pollute.
Explosives are also used in mining and oil and gas exploration, where their products are not only in the air the workers breath, but can pollute groundwater supplies.
The two main ingredients in primers for military, police, and civilian firearms are lead azide and lead styphnate. These were selected in the early 1900s to replace mercury fulminate, to eliminate the toxic effects of mercury. Unfortunately, lead is also a toxic heavy metal, and we are just now getting around to eliminating it, in turn.
Toxic lead salts at firing ranges can reach dangerous levels. One report in 1991 found that lead in the cells of people who had just cleaned an FBI firing range was over ten times above government health limits. Over a thousand pounds of lead primary explosives are used by the US Army alone each year (710 pounds of that is lead), just from the primers in small caliber ammunition. Ninety-five percent of that is used in training, meaning that the lead pollution is here at home, and primarily affects our service personnel and police officers. (Mercury fulminate is currently illegal in most of the world for use in primers and blasting caps.)
Replacing primer ingredients with safer compounds is ongoing. While it might seem to be a trivial change, one US Department of Defense study found that primers made with DDNP (diazodinitrophenol, discovered in 1858 by Peter Griess, the same person who later discovered lead styphnate) had perceptible delays (74 milliseconds) in igniting the secondary explosive in the ammunition tested, while lead-based primers in the same systems did not. These delays contributed to losses in accuracy in the weapons, and at least one misfire. The DDNP primers had more variation in peak blast pressure, and did not adequately ignite the secondary charge.
As these tests were on one batch of Russian made primers, it does not follow that DDNP is a failed explosive when used as a primer. Manufacturing variances and light loading may explain the performance. Nevertheless, simply replacing primer compositions is not a simple matter, and requires quite a bit of testing and manufacturing expertise with the new formulas. There is some evidence that the performance deficits from DDNP are not due to the explosion of the primer, but due to the effect that lead styphnate has on reducing bullet friction in the gun barrel. Without the extra lubrication the lead-based primer supplies, the performance in the actual weapon is erratic. Testing of the primers in the lab would not discover this.
Lead is not the only toxic substance in primers. Some use barium nitrate oxidizers, and some contain antimony. While the emission of heavy metal salts as fine particulates in the air during use is one issue, phasing out toxic materials throughout the supply chain is also desirable. Factories that produce the ammunition do not wish to pollute or expose their workers to toxic materials.
At the US Naval Air Warfare Center in China Lake, California, work is underway on electrically ignited primers. A mix of aluminum nanoparticles, and carbon black, with molybdenum trioxide as an oxidizer, is electrically conductive due to the first two ingredients. The goal is ignition within four milliseconds of the current being switched on. Called nano-thermites, these combinations of aluminum powder and oxides of less-reactive metals such as iron, copper, or molybdenum, are blended together so that the aluminum and the metal oxide are joined together in particles smaller than 100 nanometers. This leads to very fast reaction times.
The US Naval Surface Warfare Center, in Indian Head, Maryland, is also working with nano-thermites. Called metastable intermolecular composites (MICs), or metastable nanoenergetic composites (MNCs), the names refer to the fact that they are stable as aluminum and metal oxide, but can flip to aluminum oxide and elemental metal (releasing a lot of heat — thermite is used to melt steel). In their testing, they are using bismuth oxide, Bi2O3 as the metal oxide. It is being considered for all small and medium caliber ammunition, up to calibers of 40 millimeters.
In addition to bismuth oxide, the Indian Head researchers are also experimenting with molybdenum oxide, tungsten oxide, and copper oxide. One problem with the materials is that they are sensitive to friction and electrostatic discharge (static electric sparks). For safety, the mixture is prepared wet, and only dried once it has been pressed into the primer cap. The mixing of the aluminum and the metal oxides is done using ultrasonic mixing equipment, and the aluminum nanoparticles are coated with a protective layer so they do not spontaneously combust in air, and so they can be mixed with water.
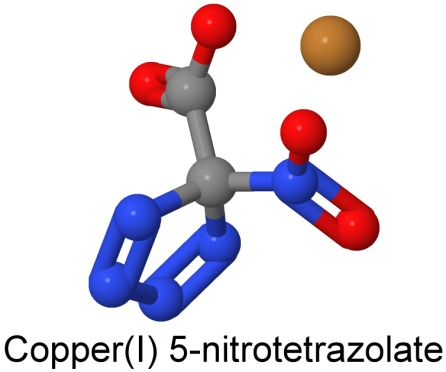
At the High Explosives Science and Technology Group, in Los Alamos, New Mexico, chemist My Hang Huynh (who won a MacArthur Foundation "Genius" grant for her work in 2007) has been working with modifications of nitrotetrazole and its salts as a primer candidate.
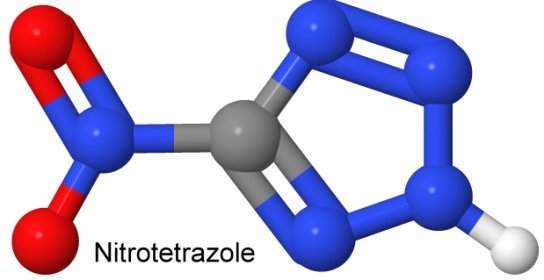
The resulting primer is insensitive when wet, an advantage from a safety point of view. Copper salts of the acid are environmentally benign. Potassium, rubidium, and cesium salts also work, but do not meet the criterion of heat stability, as they decompose before the 200° Celsius limit.
Primers are not the only problem, of course. The bullets are still lead. While shotgun loads have long been available with lead-free shot, made from bismuth (a dense, non-toxic element) or steel, replacing lead in bullets results in added costs or decreased performance. The current lead-free bullets being tested are copper-jacketed bullets filled with a mixture of tungsten and tin. Tungsten has a very high density, and the bullets can be made to have the same density as the lead bullets they replace (at a cost, however, since lead at 71 cents a pound is much cheaper than tungsten at $20 a pound). Costs are offset by the fact that metal prices are not always the gating factor in the price of ammunition, even in military quantities. Another cost saving is that the bullets can be easily recycled just by heating them. The tungsten powder is not wetted by the tin binder, and the two are easily separated.
TNT in the soil of battlefields and testing ranges is classified as a potential carcinogen. While TNT has long been disfavored in respect to nitramine-based explosives in militaries, it is still widely used in mining, for cost reasons. Mixing RDX or HMX with newer castable explosives like TNAZ results in the ability to tailor non-TNT explosives to match or surpass the performance of TNT. Again, non-polluting solutions come at a cost. TNT is used because it is cheap.
Rocket propellants that use ammonium perchlorate (such as the Space Shuttle's Solid Rocket Boosters) produce hydrogen chloride gas when burned, which becomes hydrochloric acid when it reaches water in the air or in lungs. The perchlorate itself causes thyroid problems, and has been detected in groundwater near plants that produce it. Replacing perchlorates with environmentally friendlier propellants and oxidizers is a task that has been taking years, due to the testing and formula refinement required. This will be covered in more detail in the next chapter, as will the effects of chlorine-based propellants on the ozone layer.