Some explosives, such as TNT, can be safely melted and cast into artillery shells and missile warheads. Other explosives need a little help, in the form of a small amount of an additive (generally less than 10%) that forms either a rubbery elastic material, or a hard machinable material, when mixed with the explosive.
With an explosive such as HMX, an elastomer can make the final product rubbery, and reduce the likelihood of an unplanned explosion due to shock. Fluorinated polymers (such as Teflon) are inert, and thus preferred for explosives that are chemically reactive. They can be solid in form, or they can be elastomeric. Solid forms require a less sensitive explosive, since the rigidity of the plastic may enhance the sensitivity to shock.
Some binders are explosive themselves, such as those made by nitrating a polymer. They thus serve two purposes — they bind the ingredients into shape, and contribute to the force of the explosion. Inert binders add mass that does not contribute to the explosion.
Plastic bonded explosives find use in many nuclear weapon detonation systems. An example is PBX 9407, a mix of 95% HMX, 2.5% Estane rubber, and 2.5% BDNPA-F (a nitrated plasticizer made from BDNPA and BDNPF).
Other PBX formulas use RDX, PETN, or TATB as the explosive, and binders like polystyrene plastic, nitrocellulose, polyurethane rubber, hydroxyl-terminated polybutadiene rubber, or Viton fluoropolymer elastomer.
Detonating cord made from PBX is less sensitive and more waterproof than those filled with pure PETN are. Torpedo warheads and other underwater weapons also benefit from the water resistance of PBX explosives, which are also much less sensitive to shock and impact than previous charges. They are also better explosives than the TNT and RDX they replaced.
One criterion for selecting an elastomeric binder is its glass transition temperature. It is no longer elastic, and becomes brittle below this temperature. This greatly reduces its ability to protect the explosive from shock. If the weapons are to be used or stored in cold weather, this can be a serious issue.
In shaped charges, the binder is responsible for keeping the carefully engineered form of the explosive in the right shape. If a binder is not resistant to fatigue, the shaped charge will not perform properly. Flaws and cracks also leave the explosive vulnerable to shock detonation or fragmentation.
In factories with equipment designed for melt-casting TNT, a binder that has the same properties can save money by saving the need for retooling. Likewise, equipment designed for extruding, or cold-pressing TNT can still be used if the right binder is chosen in a PBX mix.
As late as 1960 the search for a castable explosive that could withstand 500° Celsius (e.g. in a plane crash) had led to compressible compositions based on diaminotrinitrobenzene (DATB), bonded with fluorocarbon binders and pressed into shape. This mixture could survive as high as 400°. But castable explosives close to this temperature range were generally using HMX (melting point 286°), and TNT melts well below these temperatures (80°).
Casting an explosive like TATB in a thermosetting binder such as epoxy or polyester resin (or a more heat tolerant binder, such as silicone or fluoropolymers) was considered in 1967, but the characteristics that make TATB insensitive to detonation also make it insoluble in almost all solvents. As it is manufactured in the form of a fine powder, and cannot be dissolved and re-crystallized in larger form, meaning that the loading density (the ratio of explosive to binder) would be too low, lowering the detonation velocity below that of TNT.
More recent research has centered on energetic polymers (which is to say, explosives that can polymerize into long rubbery or plastic-like chains). BDNPA and BDNPF are two examples already mentioned, but more recently molecules like poly(glycidyl nitrate), called polyGlyN,
glycidyl azide polymer, called GAP,
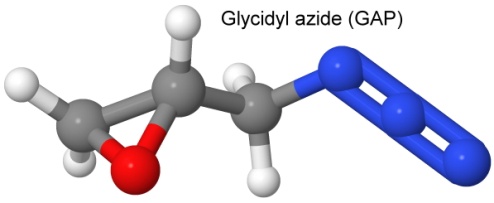
and poly(3-nitratomethyl-3-methyloxetane), called polyNIMMO are being used.
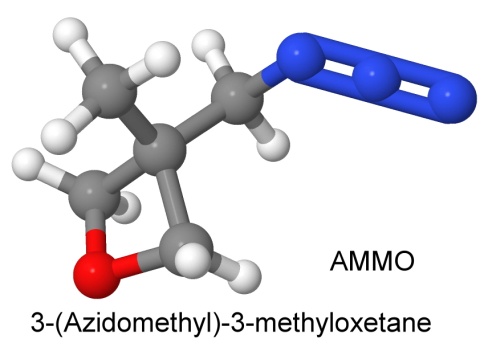
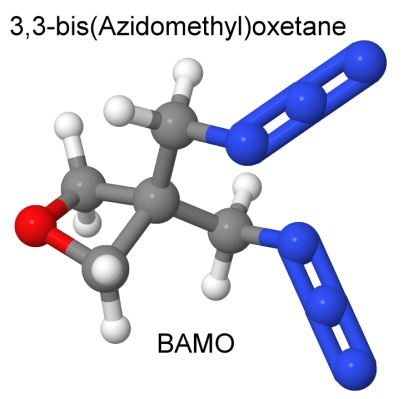
A mixed polymer of 3-azidomethyl-3-methyloxethane (called AMMO) and 3,3-bis(azidomethyl)oxetane (called BAMO) is amusingly known as AMMO/BAMO, and is used in rocket propellants, and as an energetic binder for FOX-7 explosives.
Polymer bonded explosives are frequently given names that reflect where they were developed. Those developed at the Lawrence Livermore National Laboratory have designations that start with LX- followed by a number, such as LX-09, LX-14, or LX-14-0. Those developed at the Los Alamos National Laboratory start with PBX and have four digit numbers starting with 9, such as PBX-9502. Sometimes in the literature, the hyphen is omitted or replaced by a space. Polymer bonded explosives developed by the US Navy have names that start with PBXN- and a number, such as PBXN-7. Other designations may have the initials of the laboratory in the name, such as PBXIH-18 (developed at the US Naval Surface Warfare Center, in Indian Head, Maryland), or PBXW-115 (developed at the US Naval Surface Warfare Center, White Oak, Maryland). Some extrudable explosives have an XTX- designation, such as XTX-8003.