The explosive compounds that are very sensitive were discovered first, since once you have made something that blows up in your face, you tend to notice that special property. For less sensitive explosives, it can take some time between the discovery of the substance, and the identification of its explosive potential. This is the case for many explosives, and we will see it repeatedly as we discuss more explosives. It is certainly the case with guncotton (nitrocellulose).
Sometime around the year 1310, an alchemist named Paul of Taranto published a series of books on his trade, under the name Geber, seeking to gain stature for the work by attributing it to the famous Arabian alchemist of that name. In those books, he describes the production and use of several new compounds, including nitric acid, aqua regia, sulfuric acid, and silver nitrate.
Nitric acid, especially when combined with concentrated sulfuric acid, came to be a very effective way of adding nitrate and nitro groups to organic compounds, to produce high explosives.
Henri Braconnot was a French chemist and inventor. He had learned chemistry in part by attending the lectures of comte de Fourcroy just after the turn of the 19th century, during a brief (two year) stay in Paris. Specializing in the chemistry of plants, he is known mainly for the discovery of the plant structural molecules pectin and chitin, the sugar glucose, the amino acid glycine, and several organic acids. He comes to our attention for some experiments he performed by treating plant material with acids in 1832.
The discovery of glucose came through the addition of wood, straw, and cotton to sulfuric acid. When Braconnot tried nitric acid on starch, he found he had produced a flammable substance he could dissolve in acetic acid to produce a glassy varnish. He named the substance xyloïdine. He found that sawdust, cotton, and linen produced the same substance when nitrated the same way. Today, we call the substance nitrocellulose, and we call the varnish (nitrocellulose dissolved in ether) celluloid.
Nothing much came of his discovery.
Thèophile-Jules Pelouze was another French chemist, who, despite being the son of an industrial chemist, and a laboratory assistant to Gay-Lussac, is not known for any great discoveries or inventions. But he did play with adding nitric acid to cardboard and paper, making nitrocellulose in 1838. He is mostly known today for his two students, Ascanio Sobrero, who discovered nitroglycerin, and Alfred Nobel, who tamed nitroglycerin with his invention of dynamite.
Pelouze experimented with Braconnot's xyloïdine, and noted that it ignited at 180° Celsius, burned violently, and left no residue. He experimented with nitrating paper, and produced a waterproof parchment-like material that he thought was paper coated with xyloïdine, but was in reality nitrocellulose all the way through. However, although he did his experiments believing the material would be useful in artillery, he did not commercialize his results.
A third French chemist, Jean-Baptiste Dumas, also played with adding nitric acid to cellulose, calling his creation nitramidine. But he also did nothing with it.
The main reason these three chemists ignored nitrocellulose as a propellant or an explosive is that their mixtures were highly unstable. The acid left in the fibers would continue to act on them, generating heat and sometimes premature detonation or the material would break down and lose its effectiveness. Not to mention what all that nitric acid would do to a gun barrel.
Then, in 1846, the story goes; a Swiss-German chemist named Christian Friederich Schönbein was working at home, and spilled some concentrated nitric acid on the kitchen table. Grabbing his wife's cotton apron, he mopped up the spill. As he dried the apron on the door of the stove, it flashed briefly and disappeared.
The story may be apocryphal. Schönbein had discovered ozone and published his discovery in 1840, and in 1847 published a paper describing his work with nitrating cane sugar, and implying that his work was prompted by his earlier work with ozone. To nitrate the sugar, he mixed two parts of sulfuric acid with one part of nitric acid, and stirred in finely powdered sugar in an ice bath at 0° Celsius. The thick mass that resulted was then washed thoroughly to remove the acid and then slowly dried without overheating.
The resulting nitrosucrose was brittle until warmed, but then becoming plastic, and finally runny at the boiling point of water. When heated too much, it disappeared with a flash. He then experimented with other nitrated organic substances.
The substance to which I have given in German the name of schiesswolle, and in English that of gun-cotton, having excited a lively curiosity, it may be interesting to the scientific world to become acquainted with some details of the way in which I was first led to its discovery.
The results of my researches on ozone led me in the course of the last two years to turn my attention particularly to the oxyds of nitrogen, and principally to nitric acid.
Schönbein goes on to describe his theory that hydrated sulfuric acid was a mixture of SO2 and HO2, and not SO3 and HO as the prevailing theory held. He then goes on describing the HO2 part:
My experiences with ozone having shown that this body, which I consider to be a distinct peroxyd of hydrogen, forms, as well as chlorine, at the ordinary temperature, a peculiar compound with olefiant gas, without apparently oxydizing in the least either the hydrogen or the carbon of this gas, I had the idea that it would not be impossible that certain organic matters, exposed to a low temperature, would likewise form compounds, either with the peroxyd of hydrogen alone, which, on my hypothesis, occurs in a state of combination or of mixture in the acid mixture, or with NO4.
At this point, he describes his work nitrating sugar, and then other organic substances.
In March, I sent specimens of my new compounds to some of my friends, in particular to Messrs. [Michael] Faraday, [Sir John] Herschel, and [Sir William Robert] Grove. It is necessary to note expressly that the gun-cotton formed part of these products; but I must add, that hardly was it discovered when I employed it in experiments of shooting, the success of which encouraged me to continue them.
It must have been nitrocellulose's time, since two more German chemists also made the compound in the same year.
Schönbein describes being the first to fire a cannon using the new guncotton, after testing it in smaller mortars to ensure it could safely be done. He then used it to blast some rocks and blow up some old walls, proving to himself that his invention was superior to gunpowder for those purposes. He went to England and experimented with it in Cornish mines, and with many small arms. In his paper, he is careful to distinguish his guncotton from Braconnot's xyloïdine, saying the latter had too much carbon and not enough oxygen to be a good explosive.
Washing the acid from the product was not immediately recognized as being important. In the same year as the latest discovery (1846), a plant was built in England to manufacture guncotton using English patents based on Schönbein's discovery. Due to a lack of safety precautions, the plant exploded that July, killing 21 of the workers. The company Schönbein had contracted with to produce the explosive, John Hall & Sons, refused to continue manufacturing it. Two other guncotton plants, in France, also exploded, and no more attempts to produce guncotton in either England or France were made for another 15 years.
Schönbein then contacted the Deutscher Bund, the loose confederation of 39 German states in Central Europe that replaced the former Holy Roman Empire. A committee studied his proposal for years, finally deciding to take no action. On the advice of Austria's representative to the committee, the Baron Wilhelm Freiherr Lenk von Wolfsberg, a Major General and owner of the Corps Artillery Regiment, Austria then purchased the rights to the process for a price much below what Schönbein had originally asked.
In 1853, von Lenk (as the Baron is usually referred to) developed a much more elaborate method for washing and preparing guncotton, involving washing it for three weeks, then boiling it in alkali, mixing it with sodium silicate, and then drying it. He tried his new product in iron guns, but the mixture damaged them too much. In bronze guns, which didn't shatter as easily, he had better success. He experimented with exploding shells filled with guncotton, but these often burst inside the cannon, due to the sudden acceleration provided by the new propellant. He had better luck sending the guncotton shells towards the target when he used old-fashioned black powder in the cannons.
The Austrian experiments ended in 1863, when the manufacturing plant exploded in Hirtenberg, and the Austrian government abandoned guncotton manufacture.
It took years before the British chemist Sir Frederick Augustus Abel came up with a safer way to manufacture the explosive and production resumed in 1865. Abel had studied chemistry at the Royal Polytechnic Institution, and the Royal College of Chemistry before, in 1852 at age 25 he was appointed lecturer in chemistry at the Royal Military Academy, a post previously occupied by Michael Faraday.
Abel used a pulping process to aid in the rinsing, and produced the nitrocellulose in a more useful form. Several other researchers had been experimenting with guncotton, and had established that thorough rinsing was needed, and sometimes neutralization with alkalis. Carding the resulting fibers during the rinse was also recommended. This proved to be key, as separating the fibers allowed a better rinse. In the intervening 15 years, explosions at plants in Vienna and elsewhere caused further moratoria on its manufacture.
Abel's 1865 patent notes the method of pulping:
The fibre is then taken in the wet state and converted into pulp in the same manner as is practiced by paper-makers, by putting the fibre into a cylinder, having knives revolving rapidly, working close to fixed knives.
In Abel's patent, the guncotton is not used by itself as a propellant. Instead, it is added to a form of gunpowder comprised of 65% sodium nitrate, 16% charcoal, 16% sulfur, and 3% guncotton.
Abel published a paper in the Philosophical Transactions of the Royal Society on April 10, 1866, detailing his study of the new propellant. In that paper, he describes in detail the chemistry of the compound, but also how he came to study it:
Early in 1863, by desire of the Secretary of State for War, I entered upon a detailed investigation of the manufacture of gun-cotton, the composition of the material when produced upon an extensive scale, its behaviour under circumstances favourable to its change, and other subjects relating to the chemical history of this remarkable body.
Abel credits von Lenk with his "persevering endeavors to perfect this material", but goes on to say that von Lenk's process "does not, at first sight, present any important features of novelty". Then he continues:
But the application, upon a manufacturing scale, of the system which has been pursued under Lenk's direction at Hirtenberg, brings to light several details of considerable value, the adoption of which unquestionably ensures the attainment of greater uniformity and purity of the product of manufacture than could be secured by the earlier modes of operation.
Abel takes issue with von Lenk's three-week washing period, not finding any improvement in the product over a 48-hour washing. But the main improvement is the pulping, because cotton fibers are hollow and tubular, and the acid is difficult to remove from inside the tubes. Pulping roughens the ends of the fibers, opening the tubes to the rinse water and to the later alkali bath.
I have been using the terms nitrocellulose, nitroglycerin and nitrosucrose in this book because that is what they are called in industry, and that is how the public knows them. A chemist, however, would make the distinction between a true nitro compound (like trinitrotoluene, or TNT) and these compounds, which are more properly called nitrates. Thus, we have cellulose trinitrate, glyceryl trinitrate, and sucrose trinitrate as the proper names.
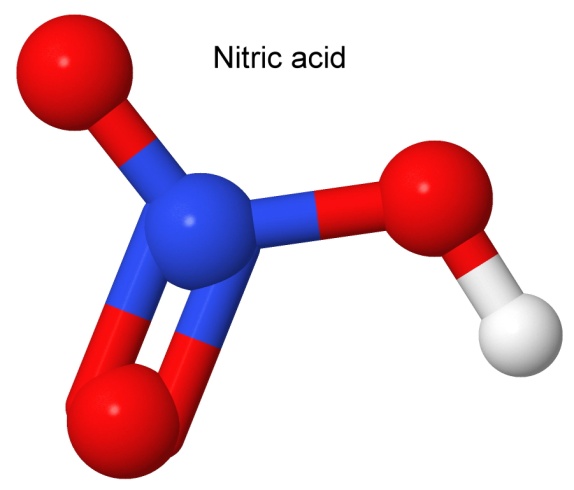
The distinction has to do with one of the oxygen atoms. In the image above, we have nitric acid. On the right side of the molecule, we have a red oxygen atom between the blue nitrogen atom and the white hydrogen atom. That oxygen atom is the difference between a nitrate group (the part of the nitric acid without the hydrogen) and a nitro group (the part of the nitric acid without the oxygen and the hydrogen on the right).
Pure guncotton was still too brisant (too shattering) to use in guns or cannon by itself. It was, however, well suited to mining, where the rock is supposed to shatter. Abel's method produced a stable form due to the removal of the acid. It did not deteriorate, and it was not affected by moisture, unlike black powder.
Another use for guncotton was in naval mines. A primary explosive (such as mercury fulminate) could detonate a small amount of dry guncotton, which could then detonate a larger charge of wet guncotton. This use of a "booster" charge would be used later with other explosives.
To make guncotton suitable as a propellant in firearms, its combustion had to be slowed. The first practical smokeless powder was invented in 1864 by a captain in the Prussian Artillery named Schultze. It was a mix of guncotton made from wood, mixed with potassium nitrate or barium nitrate. The wood contained other materials than cellulose, which, along with the nitrates, slowed the burning of the powder. Still, it was too powerful for rifles (where the bullet resists leaving the gun due to the rifling, so too much pressure builds up), but was suitable for shotguns, due to their smooth bores. Later varieties included various other adulterants to control the rate of burn.
In 1884, the French physicist Paul Vieille invented a smokeless powder that could be used in rifles. He controlled the burn rate by dissolving some of the nitrocellulose in a mixture of ether and alcohol. The remaining undissolved fibers mixed with the dissolved compound and formed a pasty jelly that could be rolled into sheets and cut, or extruded and cut like noodles.
The reason black powder produces so much smoke is that not all of the reaction products are gases. The potassium sulfides, sulfates, and oxides, along with unburned carbon from the charcoal, form a fine powder that disperses in air, and collects on the insides of the gun barrels, causing fouling. Nitrocellulose has no metals in it — what smoke it emits is primarily from unburned carbon. The reaction products are primarily nitrogen, water vapor, and oxides of carbon.
Smokeless powder had other advantages other than being smokeless. Less of it was needed, since it was more powerful than black powder. To get the same effect in a gun, only about a third as much smokeless powder is needed as black powder. Moreover, smokeless powder was far less affected by moisture.
Further improvements to smokeless powder involved coating the extruded pellets with graphite. This slowed the burn rate a bit, but just as importantly, it made the powder electrically conductive, so static electricity would not build up and create sparks during transport and handling. The extruded material allowed manufacturers to control the surface area to volume ratio, which gave them precise control over the burn rate.
While guncotton is far less sensitive to moisture than black powder is, it is still important to control how it reacts to humidity. Consistent performance as a propellant is necessary if the projectiles are going to hit their mark reliably, and how much moisture is in the powder has an effect on its performance. Guncotton was treated with hot water to create cracks in the outer skin of the colloidal extrusions, where water could collect and remain. If the grains were too dry, they would absorb moisture from the air. If they were too wet, they would dry out in transport. With the minute cracks in the surface, there was just the right amount of water, so the powder neither lost nor gained moisture during storage and transport.
Controlling the rate of burning is very important. The different extruded shapes of the grains are designed to have specific effects on the rate of burn. Simple cylinders, like that of cordite (named because it was extruded in strings or cords), have surfaces that get smaller as the grains burn from the outside in. This means that the rate of burning decreases as the charge burns.
If extruded into strips, flakes, or tubes, the powder burns at a more consistent rate. To get the powder to burn faster as the burn progresses, the grains can be extruded with multiple hollow tubes inside. In a gun, as the bullet travels down the barrel and the volume behind it increases, a steadily increasing pressure caused by faster and faster burning keeps the pressure constant. This leads to faster velocities for the projectile.
The flash of a gun at night can give away its position. A 12-inch gun can project a muzzle flash as far as 150 feet, and this can be seen reflecting off low clouds for 30 miles, farther than the sound of the gun carries.
The flash is caused by superheated air, and by hot uncombusted gases igniting when they encounter oxygen in the air. To reduce the flash, so-called flashless powders were developed. (The use of the term "powder" has continued, despite the extruded macaroni-like form of the new propellants.)
To reduce the temperature of the escaping gas, explosives that burn cooler can be added. Ammonium nitrate is one example, as is guanidinium nitrate and nitroguanidine. To prevent the gases from igniting, salts such as potassium chloride can be added that prevent the gases from combining with oxygen while they are hot enough to ignite. Adding inert material to the mixture that absorb heat and produce gas, such as carbonates that produce carbon dioxide when heated, can cool the output at the same time they increase the amount of gas produced.