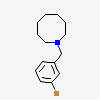OXYGEN MOLECULE
 Oxygen - definition from Biology-Online.org
Oxygen - definition from Biology-Online.org
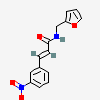
[1. (Science: chemistry) A colourless, tasteless, odorless, gaseous element occurring in the free state in the atmosphere, of which it forms about 23 per cent by weight and about 21 per cent by volume, being slightly heavier than nitrogen. symbol O. atomic weight 15.96. It occurs combined in immense quantities, forming eight ninths by weight of water, and probably one half by weight of the entire solid crust of the globe, being an ingredient of silica, the silicates, sulphates, carbonates, nitrates, etc. Oxygen combines with all elements (except fluorine), forming oxides, bases, oxyacid anhydrides, etc, the process in general being called oxidation, of which combustion is only an intense modification. at ordinary temperatures with most substances it is moderately active, but at higher temperatures it is one of the most violent and powerful chemical agents known. It is indispensable in respiration, and in general is the most universally active and efficient element. It may be prepared in the pure state by heating potassium chlorate. This

|

|

|

|

|

|
|
|
|
|
|
|
|
|
|
|
|
|
|

|
|