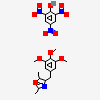
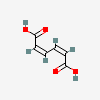
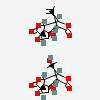Picrotoxinum
 PubChem Notes:
PubChem Notes:
Picrotoxin A noncompetitive antagonist at GABA-A receptors and thus a convulsant. Picrotoxin blocks the GAMMA-AMINOBUTYRIC ACID-activated chloride ionophore. Although it is most often used as a research tool, it has been used as a CNS stimulant and an antidote in poisoning by CNS depressants, especially the barbiturates.
Molecular Formula:
C30H34O13
InChI: InChI=1/C15H18O7.C15H16O6/c1-12(2,18)6-7-10(16)20-8(6)9-13(3)14(7,19)4-5-15(13,22-5)11(17)21-9;1-5(2)7-8-11(16)19-9(7)10-13(3)14(8,18)4-6-15(13,21-6)12(17)20-10/h5-9,18-19H,4H2,1-3H3;6-10,18H,1,4H2,2-3H3/t5-,6+,7u,8?,9-,13-,14-,15+;6?,7-,8u,9?,10+,13+,14+,15-/m10/s1
InChIKey: InChIKey=VJKUPQSHOVKBCO-IQTYKTTQBT
SMILES: CC(=C)C1C2C3C4(C(C1C(=O)O2)(CC5C4(O5)C(=O)O3)O)C.CC12C3C4C(C(C1(CC5C2(O5)C(=O)O3)O)C(=O)O4)C(C)(C)O
Names:
Caswell No. 663A
Cocculin
Cocculus
Fish berry
Indian berry
Oriental berry
PICROTOXIN
Picrotox
Picrotoxine
Picrotoxinum
Registries:
PubChem CID 31304
PubChem ID 173365
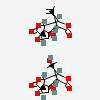
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|