Ingredients --
Nitrous oxide
Chemical Formula: N2O
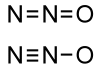
bonds resonate between single/triple and double
Synonyms
dinitrogen oxide,dinitrogen monoxide
Description
Colorless non-flammable gas with a sweet odorUses
Nitrous oxide is used as a foaming agent and propellant in whipped cream. It dissolves easily in fats under pressure, and comes out of solution when the pressure is reduced, much like the carbon dioxide in a can of soda. Many gasses will dissolve in fats, and make good propellants. However, most are flammable or toxic, or react with the fats. Nitrous oxide dissolves in the fats that sheath the nerve cells, and produces numbing and mild intoxication. It is the "laughing gas" used by dentists to make patients less aware of pain. Other possible propellants, such as the propane used in hair sprays, or freon, also cause intoxication when they dissolve in the fats around nerve cells, and so are not used, since their flammability, safety, cost, or taste makes them less desirable than nitrous oxide for whipping cream.nitrous oxide: InChI=1/N2O/c1-2-3
By Simon Quellen Field