Ingredients --
Glycerine
Chemical Formula:
Glycerine
Synonyms
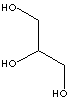
GlycerolGlycerin
1,2,3 Trihydroxy propane
1,2,3 propanetriol
Description
Glycerine is a somewhat viscous clear oily liquid, with a sweet taste. It is manufactured from petroleum, or from glycerides in fats, often as a by product in the production of soap.Uses
Glycerine is hydrophilic (water loving), and is used to keep products such as dried fruits from getting too dry, and to keep the liquid in soft fillings from escaping into crisp crusts, making them soggy. This emollient (moisturizing) effect also finds use in hand creams. It lowers the freezing point of water, and is used as an anti-freeze. Glycerine is about ¾ as sweet as sugar, so it can reduce the need for sugar in products that require it for other reasons. In toothpaste it sweetens while keeping the paste from drying when the cap is left off. Glycerine is used in many skin creams and medicines, such as eye and ear drops, poison ivy creams, suppositories, and contraceptive jellies. Glycerine is used as an emollient (skin softener) in soaps. A familiar compound results when glycerine reacts with nitric acid in the presence of sulfuric acid as a drying agent.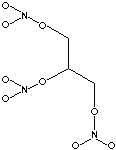
Glyceryl trinitrate Glyceryl trinitrate is used in medicines for its effects on blood flow. As a coronary vasodilator, it is used by heart patients. As an explosive, it is the principle ingredient in dynamite, under the name nitroglycerine.
glycerin: InChI=1/C3H8O3/c4-1-3(6)2-5/h3-6H,1-2H2
glyceryl trinitrate: InChI=1/C3H5N3O9/c7-4(8)13-1-3(15-6(11)12)2-14-5(9)10/h3H,1-2H2
By Simon Quellen Field