Ingredients --
Fumaric acid
Chemical Formula:
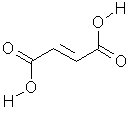
Synonyms
allomaleic acid,2-butenedioic acid,
trans-butenedioic acid,
boletic acid,
lichenic acid,
trans-1,2-ethenedicarboxylic acid
Description
Colorless crystals or white powder.Uses
Fumaric acid is used as a flavoring, because it is the sourest tasting of the organic acids. Three parts of fumaric acid are as sour as five parts of citric acid. It is also used as an anti-oxidant, as a mordant (a substance that helps dyes stick to fabric), and as a buffering agent (helps maintain a particular acidity or alkalinity). Fumaric acid is used to lower the pH (make something more acid, and thus taste more sour). This helps certain anti-microbial agents such as sodium benzoate and calcium propionate work better. Fumaric acid itself kills bacteria also. Fumaric acid breaks the sulfur-to-sulfur bonds in the elastic protein gluten in bread doughs. This makes the doughs more machineable. It also is uses in rye and sourdough breads to make them more sour. Fumaric acid is used in combination with sodium bicarbonate to create delayed action leavening agents (something that produces carbin dioxide gas to make breads rise). Since it only dissolves in warm water, the leavening action is delayed until the bread starts to bake. Because fumaric acid is not very soluble in water, it can replace hygroscopic acids in dry mixes, and thus help keep them from caking in humid conditions.fumaric acid: InChI=1/C4H4O4/c5-3(6)1-2-4(7)8/h1-2H,(H,5,6)(H,7,8)/b2-1+/f/h5,7H
By Simon Quellen Field