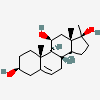
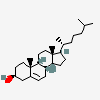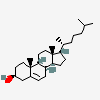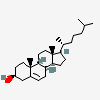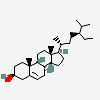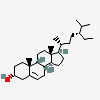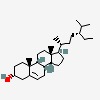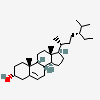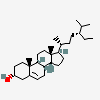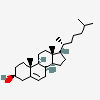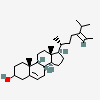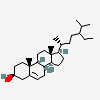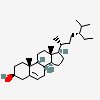Hermaphrodiol
 PubChem Notes:
PubChem Notes:
Androstenediol An intermediate in TESTOSTERONE biosynthesis, found in the TESTIS or the ADRENAL. Androstenediol, derived from DEHYDROEPIANDROSTERONE by the reduction of the 17-keto group (17-HYDROXYSTEROID DEHYDROGENASES), is converted to TESTOSTERONE by the oxidation of the 3-beta hydroxyl group to a 3-keto group (3-HYDROXYSTEROID DEHYDROGENASE).
Molecular Formula:
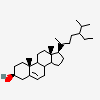
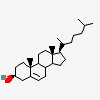
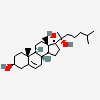
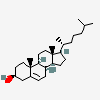
C19H30O2
InChI: InChI=1/C19H30O2/c1-18-9-7-13(20)11-12(18)3-4-14-15-5-6-17(21)19(15,2)10-8-16(14)18/h3,13-17,20-21H,4-11H2,1-2H3/t13-,14-,15-,16-,17-,18-,19-/m0/s1
InChIKey: InChIKey=QADHLRWLCPCEKT-LOVVWNRFBV
SMILES: [H][C@@]12CC=C3C[C@@H](O)CC[C@]3(C)[C@@]1([H])CC[C@]4(C)[C@@H](O)CC[C@@]24[H]
Names:
Androstenediol
Androst-5-enediol
Androst-5-ene-3beta,17beta-diol
androst-5-ene-3beta,17beta-diol
Hermaphrodiol
ZINC03814414
(3S,8S,9S,10R,13S,14S,17S)-10,13-dimethyl-2,3,4,7,8,9,11,12,14,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthrene-3,17-diol
3beta,17beta-Dihydroxyandrost-5-ene
3beta,17beta-Dihydroxy-5-androstene
521-17-5
Registries:
PubChem CID 10634
ChEBI 2710
Kegg C04295
PubChem ID 12146100
PubChem ID 6955
|
|
|
|
|
|
|

|
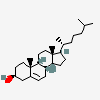
|
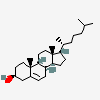
|
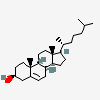
|
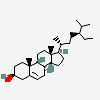
|
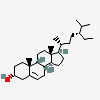
|
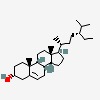
|
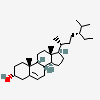
|
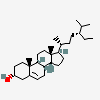
|
|
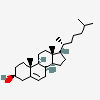
|
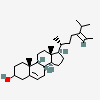
|
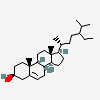
|
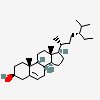
|